The main goal of our lab is to regulate two aspects of mitochondria, A)
energy production and generation of reactive oxygen species, and B) cell
death, that benefit human health.
- Background and Our Mission:
Here is why we are studying mitochondria and what kind of research we are working on. - 1) Mitochondria are a double-edged sword. (energy production and generation of reactive oxygen species)
- 2) Mitochondria relay cell death signals.
- 3) What can be found out inside the cell and what can be found out by taking it out.
- 4) What can be found out from single mitochondria.
- Research Projects and Main Achievements:
The following is a description of the main results of our research to date. - 1) Development of techniques to measure and manipulate intracellular organelles
- 2) Research on how mitochondria work
- 3) Research on the effects of mitochondria on cells
[Background and Our Mission]
1) Mitochondria are a double-edged sword.
More than 2 billion years ago, aerobic bacteria, alpha proteobacteria (ancestors
of mitochondria), came to work inside cells. As a result, cells were able
to use oxygen to produce a large amount of energy, and organisms evolved
significantly. This is why mitochondria are thought to have been the driving
force behind evolution. On the other hand, it is not only a good thing,
but at the same time the cells have taken on the danger of utilizing highly
reactive oxygen inside the cells.
This double-edged sword nature of mitochondria is still carried on into
all organisms that have mitochondria, including humans. Damage caused by
reactive oxygen species generated in mitochondria may not only appear rapidly,
but also gradually accumulates and affects us. In the short-lived and nutrient-deficient
era that our ancestors once experienced, the advantages of mitochondria
were emphasized and the disadvantages were not so obvious. However, in
today's age of abundant nutrition and longevity, the negative effects of
mitochondria can no longer be ignored. In fact, the diseases that caused
by or associated with mitochondrial oxygen hazards are very diverse, including
neurodegenerative diseases. We are studying to find ways to regulate mitochondria
that are appropriate for an age of nutrient-rich and aging population,
and to contribute to people today and in the future.
2) Mitochondria relay cell death signals.
Our cells have a mechanism called programmed cell death, in which intracellular
proteins work together to cause the cell to die. For example, programmed
cell death called apoptosis kills cancer cells and prevents them from proliferating.
On the other hand, if apoptosis occurs in a hypersensitive manner, cells
that do not need to die will die, leading to early signs of aging, for
example. As in this example, proper regulation of cell death is extremely
important for living organisms. Mitochondria act as relay points for the
cell death signals as it transmit through the cell during apoptosis and
parthanatos, two of the programmed cell deaths. We would like to investigate
how mitochondria work during cell death and find ways to control cell death.
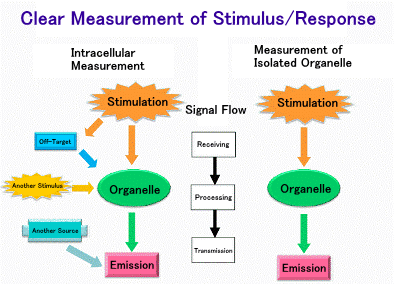
3) What can be found out inside the cell and what can be found out by taking
it out.
There are two ways to study the functions of mitochondria: one is to examine
their behavior inside the cell, and the other is to take them out of the
cell and examine their mechanisms. Both approaches are necessary for an
accurate understanding. The purpose of examining mitochondria inside the
cell is to understand how they behave inside the cell. On the other hand,
the purpose of extracellular investigation is to strictly regulate the
environment around mitochondria and investigate the mechanisms by which
the behavior observed inside the cell occurs. These are illustrated in
the figure below. We are developing both methods.
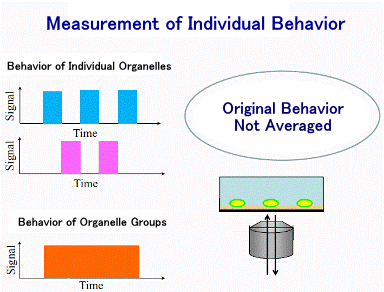
4) What can be found out from single mitochondria.
In previous studies on mitochondria taken out of the cells (mechanism studies),
We were observing the sum of signals emitted from a large number (millions
to hundreds of millions) of mitochondria suspended in buffer solution.
In this situation, the changes in individual mitochondria are averaged
out, and fluctuations such as repeated increases and decreases cannot be
seen. Therefore, it is quite possible that we are missing the exact behavior
of mitochondria and are not approaching the original mechanism. Based on
this perspective, we adsorb mitochondria onto cover glass so that we can
observe the behavior of each individual mitochondria using a microscope.
【Research Projects and Main Achievements】 ←Currently being revised
1) Development of technology to measure and manipulate mitochondria
In order to manipulate mitochondria as intended, we are developing both
techniques for manipulating mitochondria and techniques for detecting changes
caused by manipulation. Below are some specific examples.
A) Method to isolate mitochondria without damaging them (Okutani et al.,
2025)
Mitochondria have a structure in which the outer membrane surrounds the inner membrane, and this structure is critical to mitochondrial function. While mitochondria extracted from cells (isolated mitochondria) are valuable samples, it has been extremely difficult to obtain a population of isolated mitochondria in which the majority retain this outer-membrane-surrounding-inner-membrane structure.
The reasons are as follows. This is because significant damage was inflicted
on the mitochondrial outer membrane when cells were homogenized (mechanically
crushed and homogenized) or when cell membranes were dissolved using surfactants
to extract mitochondria. If the entire cell is crushed, the mitochondria
are naturally crushed as well; similarly, if the cell membrane is solubilized,
it is reasonable to assume that the mitochondrial outer membrane will also
be solubilized immediately thereafter.
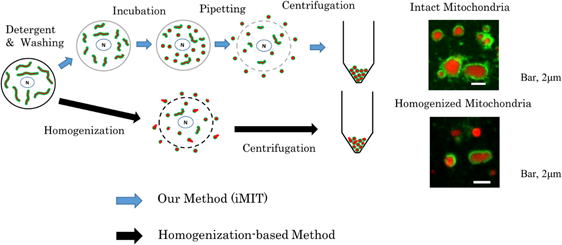
Therefore, we developed a method (iMIT) to obtain a mitochondrial population
consisting predominantly of mitochondria in which the outer membrane surrounds
the inner membrane, without homogenizing or solubilizing the cells.
The iMIT method consists of the following four steps (see figure below).
1) Selectively weaken the cell membrane. See “Detergent & Washing”
in the figure below.
It is important that no holes are created in the cell membrane during this process.
2) Contract the mitochondria. See “Incubation” in the figure below.
This makes it easier to extract the mitochondria.
3) Selectively rupture the cell membrane. (See “Pipetting” in the figure
below)
A gentle water jet is applied to selectively create holes in the cell membrane.
4) Collect the mitochondria that have leaked out of the cells. (See “Centrifugation” in the figure below)
For details, please refer to the published paper (Okutani et al., 2025).
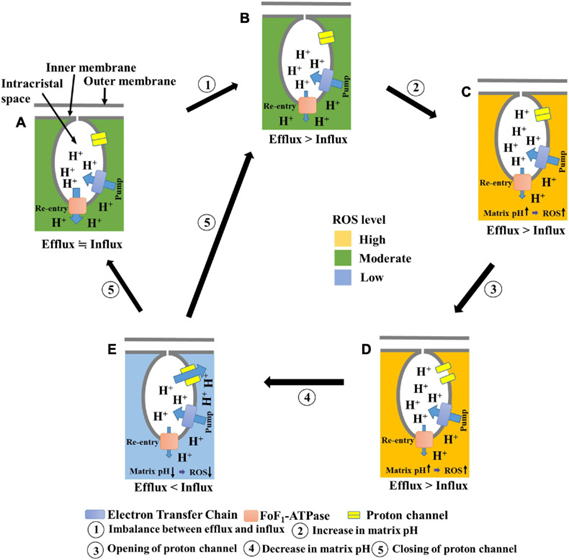
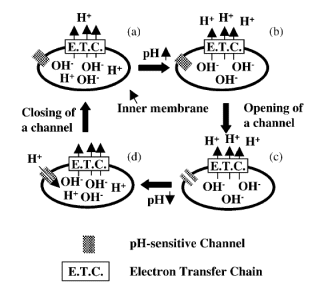
B) The mechanism by which mitochondria undergo temporary depolarization
(Aklima et al., 2021)
We quantitatively measured membrane potential fluctuations in a single
mitochondrion and discovered a mechanism that electrically short-circuits
mitochondria. This discovery indicates that mitochondria reduce reactive
oxygen species production without decreasing ATP levels, the energy currency
within cells.
2) Study on how mitochondria work
In order to control the mitochondria as intended, we need to know what
and how to control them.
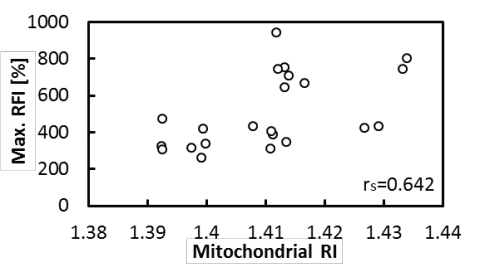
A) Simultaneous measurement of mitochondrial internal density and activity
(Haseda et al., 2015)
We have developed a method to measure the refractive index of a single
mitochondrion and discovered that mitochondria become polarized when the
internal density of mitochondria increases. This finding suggests that
mitochondrial expansion and contraction occurring within the cell may be
an activity regulation mechanism.
B) Mechanism of temporary depolarization of mitochondria (Hattori et al.,
2005)
We quantitatively measured membrane potential fluctuations in a single
mitochondrion and discovered a mechanism that causes mitochondria to electrically
short the mitochondria. This finding is expected to lead to the suppression
of reactive oxygen generation by mitochondria.
3) Study of Mitochondrial Effects on Cells
A) Recovery of Cells Damaged by Oxidative Stress through Mitochondrial
Supplementation (Ibban et al., 2025)
Adding mitochondria to hydrogen peroxide-damaged cardiomyocytes (H9c2)
facilitates recovery from damage. This effect is particularly pronounced
with the aforementioned intact mitochondria, raising hopes for new medical
applications.
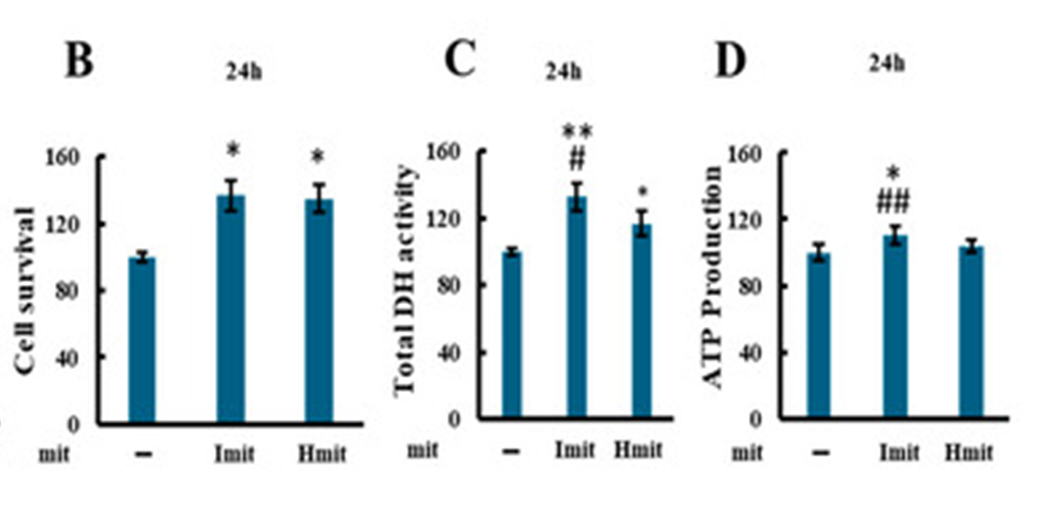
The A–C panels above compare cellular recovery when mitochondria are not
added to cells damaged by oxidation (–) versus when mitochondria are added
(Imit denotes intact mitochondria, Hmit denotes mitochondria with damaged
outer membranes).