C(sp3)–H bond functionalization by internal redox system
The multi-step sequence was required for the synthesis of fine chemicals (such as drugs, agrochemicals). Substantial amount of organic (inorganic) wastes were produced in the process. Thus, the development of environmentally benign process was strongly demanded. C–H bond functionalization reaction is the highly promising approach to satisfy this demands. The transformation of inert C(sp3)–H bond was difficult task, even though the extensive studies for the C–H bond functionalization reaction have been conducted. Furthermore, the employment of the toxic and expensive transition metal and excess amount of external oxidants was required to accomplish this transformation in most cases.
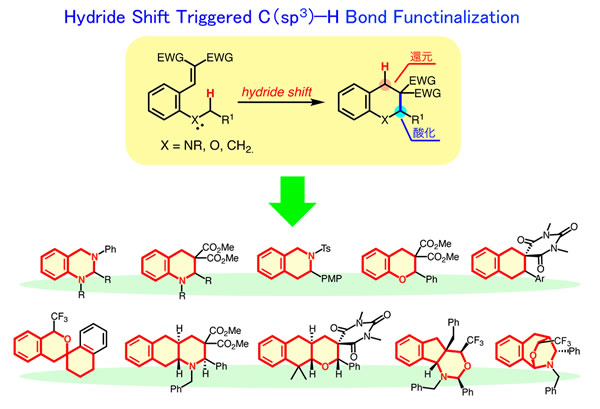
Recently, we have developed novel C(sp3)–H bond functionalization strategy, hydride shift/cyclization system (internal redox system). There are three key features in the reaction: (1) direct transformation of inert C(sp3)–H bond, (2) catalytic reaction with simple acids (Brønsted or Lewis acids), and (3) construction of fused-polycyclic skeleton by single operation. Our future plan is the synthesis of complicated polycyclic framework and some biologically active compounds based on this method.
Development of efficient synthetic method by novel catalytic system
※Under construction


